A collaborative project between ESR and Sysmex New Zealand has been able to streamline vital data on COVID-19 testing to the Government.
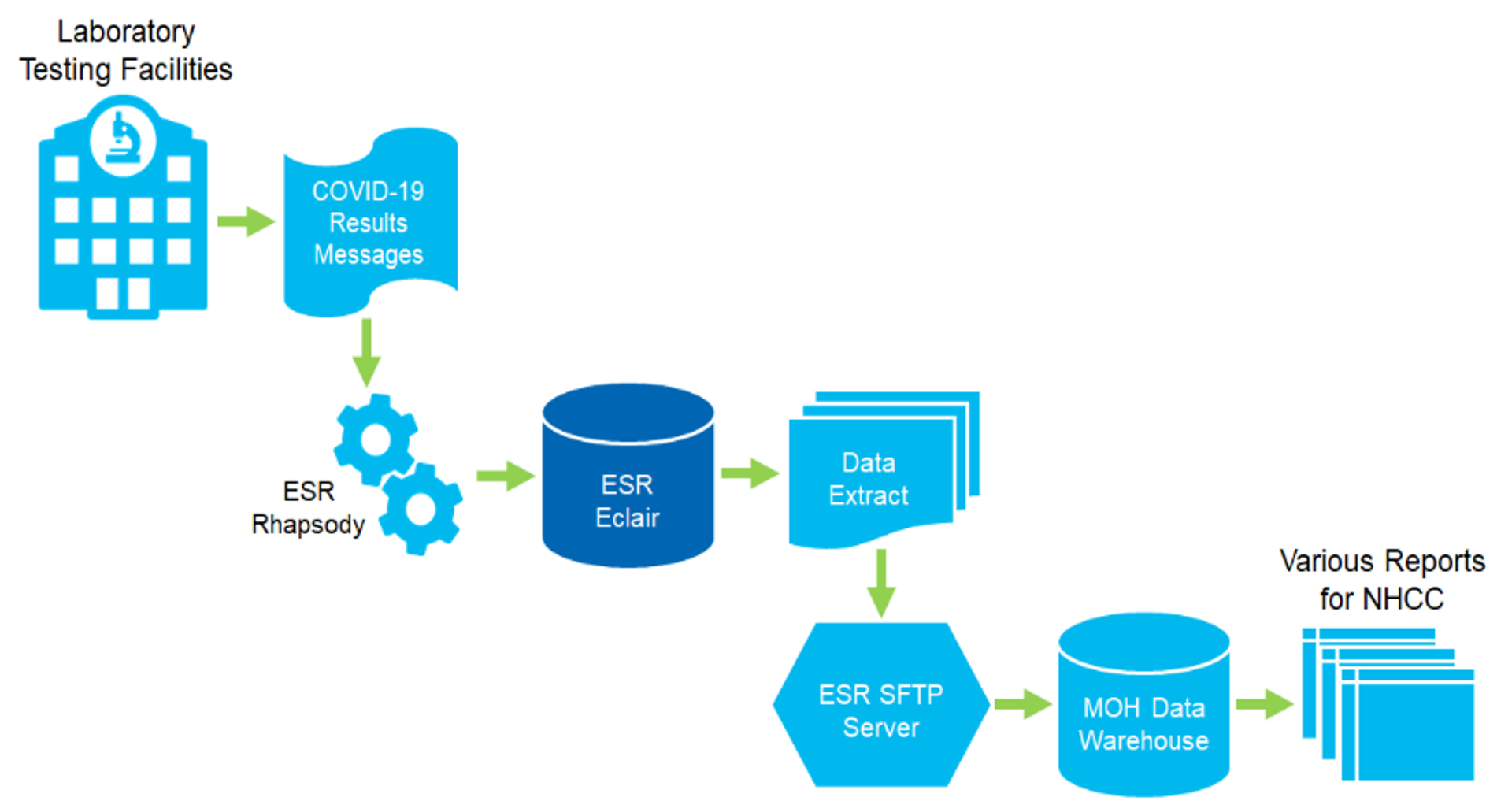
ESR has worked with Sysmex to establish a national clinical data repository (CDR) for all COVID-19 test reports from all testing lab facilities across the country. The repository complements and enhances the national notifiable disease surveillance system that ESR operates on behalf of the Ministry of Health.
The CDR will act as a repository for all test results, both positive and negative, from all health laboratory testing facilities in New Zealand. The CDR will make it easier, quicker and more efficient for information to be made available for the intelligence reporting that ESR provides to the National Health Coordination Centre (NHCC) for the Ministry to use in the daily COVID-19 update to the nation.
ESR Data and Informatics Manager Andrew Crooke says the system allows a complete national overview of all testing for the Ministry of Health to be provided much faster.
“COVID-19 has produced unique challenges. The speed at which data is needed at the moment is unprecedented and we wanted to find a way to streamline processes for the laboratories, ourselves and the Ministry of Health. So, we teamed up with Sysmex New Zealand and every laboratory in the country to find a solution.
“As much as Sysmex has risen to the challenge, we must also thank the laboratories who have been working with us to meet this challenge and implement the system. Their work has been outstanding as we move forward with this solution.”
Sysmex’s Eclair CDR is a proven and trusted solution for the standardised storage, management and display of diagnostic and clinical data by District Health Boards and private pathology providers across New Zealand and overseas.